[Microbubble] How to distinguish different microbubble generation technologies
How to distinguish between different microbubble generator technologies.
Method of judging the superiority / inferiority of the many microbubble generators glutting the market.
There are various types of micro/nano bubble generators on the market. It is difficult to determine which products match the purpose of your use. We will explain how to judge superiority / inferiority of micro/nano bubble generators.
How to distinguish (1) : Can microbubbles be generated in pure water? How about in organic solvents?
Ask the manufactures of microbubble generators the question “Is your technology able to generate microbubbles in pure water?
How about in organic solvents?” This question is valid to judge their technical level. Nearly all technologies would have to
answer ‘NO’ to this question. This means they are not suitable for accelerating chemical reactions.
In order to generate micro/nano bubbles in pure water or in organic solvents, it is necessary for the microbubble generator to be
able to strongly break down and mix both gas and liquid. In other words, the technological level of gas-liquid reaction
of the microbubble generator can be determined roughly by this question alone.
It is known that microbubbles are easily made in water containing many ionizing substances or impurities.
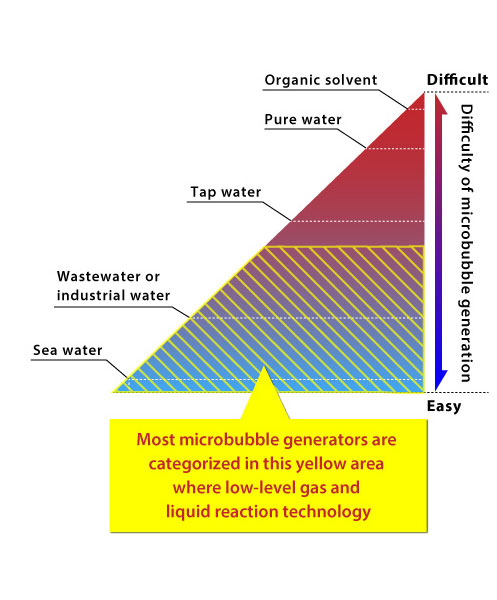
- Sea watercontains many varied ionized minerals, so microbubbles are very easy to make unlike in fresh water.
- Sea water contains many varied ionized minerals, so microbubbles are very easy to make unlike in fresh water.
- In wastewater or industrial water, microbubbles are easy to make.
- Dissolved gases are pulled in by the impurities contained in the water, and they change easily into bubbles.
- In tap water, microbubbles are difficult to make.
- There are many generators that can not make microbubbles in tap water. Therefore, in order to make microbubbles easily, there are companies that secretly use salt water in their demonstrations in exhibition halls. Please be careful.
- In pure water, most technologies can not generate microbubbles.
- This is because pure water contains very few ionized substances.
- In organic solvents, there are almost no technologies that can generate microbubbles.
- Gases are easy to dissolve in organic solvents because density / specific gravity / viscosity are small.
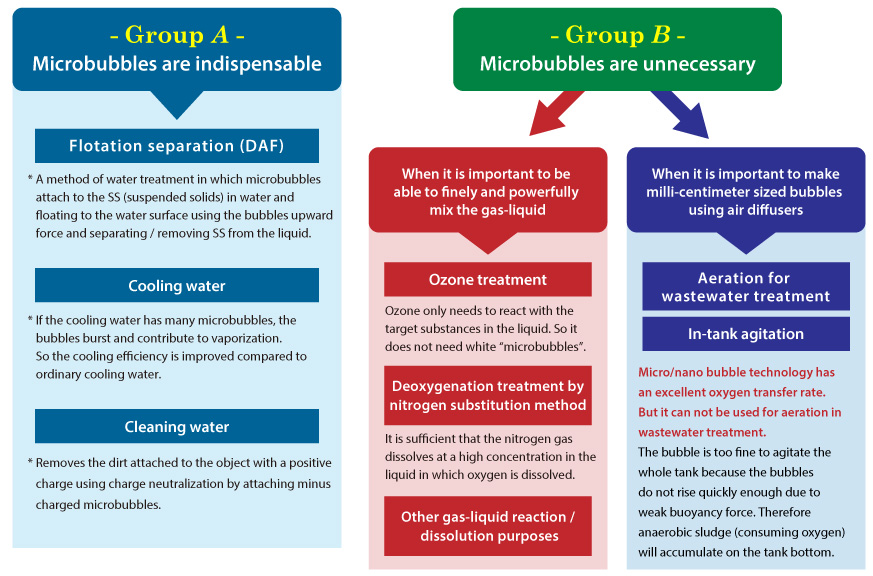
How to distinguish (2): Are transparent bubbles superior to cloudy bubbles?
Some people claim that cloudy bubbles “microbubbles” are less effective than transparent bubbles “nanobubbles”. But this is incorrect.
Depending on the purpose of the gas-liquid reaction, two categories can be identified. In group “A” cloudy bubbles (microbubbles) are necessary. In group “B” cloudy bubbles are not necessary and nano-sized bubbles or coarse bubbles are effective".
Therefore, although the optimum bubble size varies depending on the purpose, the claim that "finer bubbles are superior" is simply not true.
How to distinguish (3): Look at the actual performance records in detail
Oxygen gas, nitrogen gas, hydrogen gas, ozone gas are typical gases that are difficult to dissolve in liquid. So, in order to increase the efficiency of chemical reaction using these gases, it is necessary to break down these gases to micro/nano bubbles and dissolve/react them. Micro/nano bubble technology can instantly increase the dissolved gas concentration to oversaturation point but it requires much more energy than using conventional air diffusers. Therefore micro/nano bubble technology is suitable for applications where it necessary to dissolve a small amount of gas with high efficiency (quality rather than quantity). To avoid wasting a lot of energy and money, a sober analysis is necessary.
For example, micro/nano bubble technology is effective at dissolving nitrogen gas in order to remove dissolved oxygen from liquid (deoxygenation by substitution with nitrogen gas), as it is necessary to dissolve nitrogen to the oversaturation point in the liquid instantly.
But you should NOT use this technology to increase DO (dissolved oxygen) value in a large volume of low oxygen water (e.g. wastewater, pond and lake water) because in these situations a large volume of oxygen is needed. Gas quantity not quality is important. Also, easily dissolving gas (e.g. carbon dioxide gas, hydrogen sulfide gas, chlorine gas) is not suitable for use in micro/nano bubble technology. For example drinks manufacturers do not use micro/nano bubbles to carbonate their beverages.
Coarse bubbles are sufficient because carbon dioxide gas is very easy to dissolve into liquid. (*However in some cases micro/nano bubble technology is suitable to make carbonated drinks. For details, please contact us.)
Most micro/nano bubble manufacturers claim with supporting records that their technology is innovative and advanced. However, these supporting records often show situations where it is unnecessary to use micro/nano bubble technology. For example they claim, “We increased the DO value of wastewater in an aeration tank”. However in reality micro/nano bubble technology is unnecessary for this situation because oxygen is easily dissolved into wastewater (which has low DO value), and coarse bubbles generated by air diffusers are more than adequate. (For deitals, see the following link.)
Micro/nano bubble technology is not suitable for aeration in aeration tank
In addition these technologies do not have a strong mixing function. Therefore they can only be used in very few accelerating chemical reaction situations.
How to distinguish (4): Difference in supersaturation level
You can dissolve gas up to oversaturation levels when you use micro/nano bubble technology.
But there are differences in the level of oversaturation depending on the technologies. For example testing the oversaturation value under atmospheric pressure (not pressurized), the saturation value is set at 100%, Company “A” technology can raise it only up to 104% oversaturation maximum, but our technology can raise it to 131%. This means there is a big difference in the ability of the different micro/nano bubble technologies to dissolve gas into liquid.
How to distinguish (5): Other criteria
(1) How many seconds will it take for the cloudy microbubbles to rise and disappear after they have been generated?
(Retention time of microbubbles)
Company X the biggest water treatment engineering company in Japan analysed and collected data from three companies; company K, company N and OHR. Microbubbled water, generated by the equipment of these three companies, was collected in 1ℓ measuring cylinders respectively to observe how long it took for all microbubbles to disappear.
The results were company K was 90 seconds, company N was 75 seconds, and our company (OHR) had a retention time of 170 seconds. The longer the retention time, the smaller the microbubble size. Among the so-called “microbubble generators”, there is a big difference between the generated microbubble qualities.
(2) How long will it take for dissolved gases in water to decrease?
The oxygen water produced by Company O has an oxygen gas concentration of 40mg/L immediately after opening the bottle, but it decreased to 20mg/L after 2 hours. Oxygen water produced by our micro/nanobubble technology remained at 36mg/L even after 2 hours. This means that there is a big difference in the gas dissolving capabilities of the gas dissolving technologies.
You are at risk of being deceived unless you carefully determine the micro/nano bubble generation technology that matches your specific requirements.













